
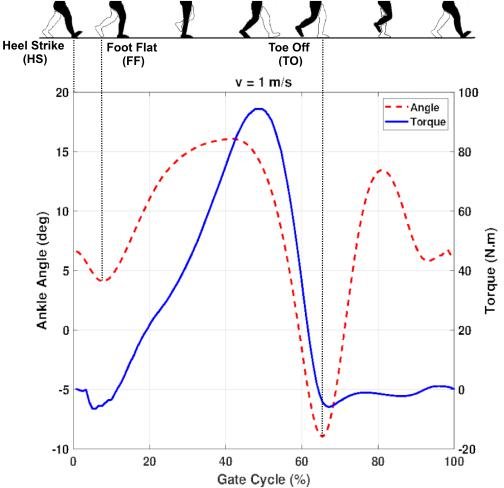
THE PROBLEM: The Mobility-Weight Paradox
In motorized lower-limb prosthetics, patient comfort is dictated by a brutal trade-off: battery life versus weight. Traditional power systems for foot-ankle prostheses were often too heavy (causing gait imbalance) or inefficient (causing thermal safety risks and frequent recharging).
To achieve clinical viability for the BioApps RoMicP®, we needed a power architecture that could support 5,000+ daily steps while maintaining a total system mass under 2.5kg.
THE SOLUTION: GaN-Based Power Architecture & Simulation
As the lead Research Engineer, I architected a high-density power conversion system centered on Gallium Nitride (GaN) technology. Using MATLAB/Simulink and Texas Instruments’ LMG series GaN FETs, I designed and simulated a three-phase inverter system.
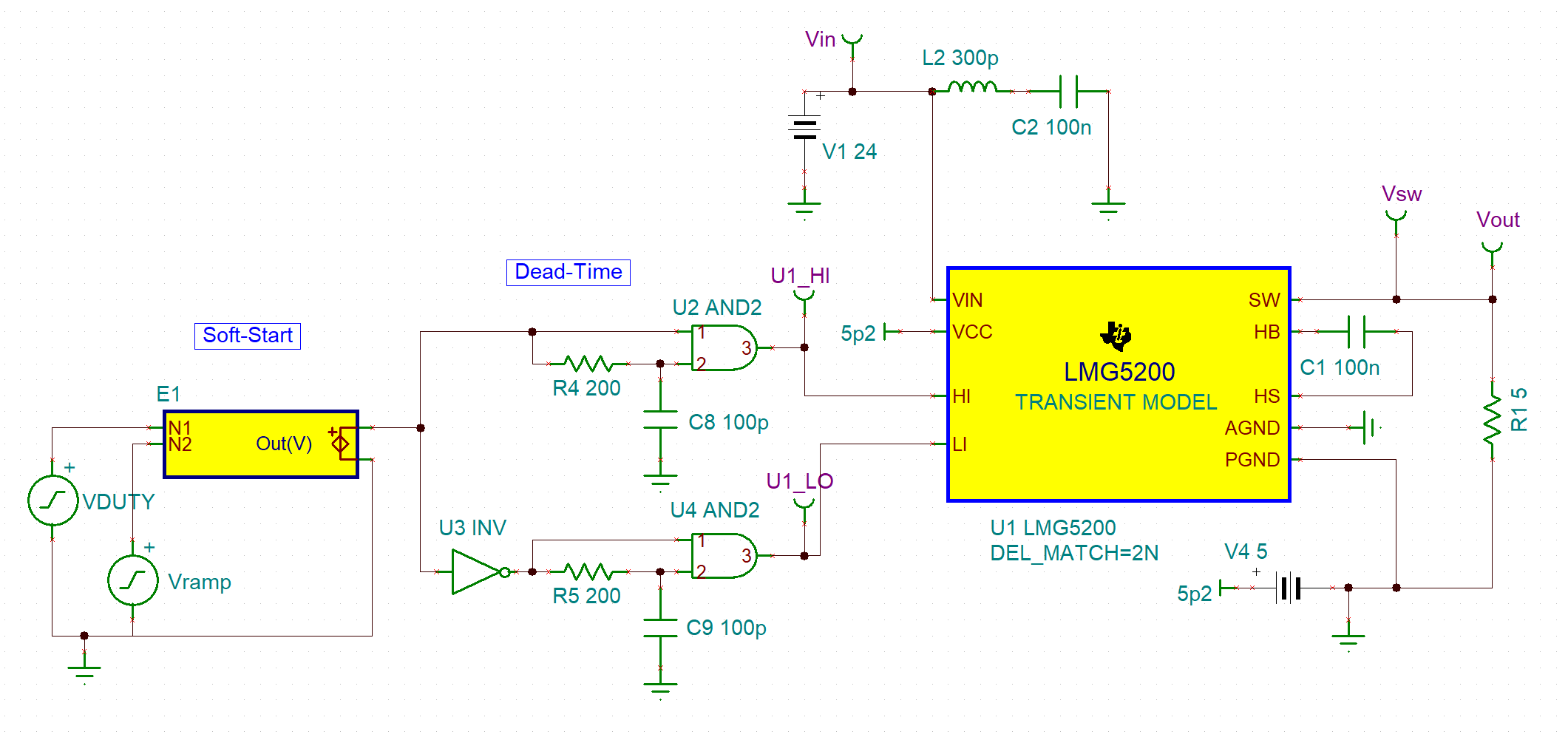
Design Choice
I performed a comparative analysis between standard PWM switching and multi-level topologies. While multi-level designs offered lower harmonic distortion (0.82%), my analysis proved they dropped efficiency to an unacceptable 59%.
Optimization
I pivoted to a high-frequency PWM switching strategy to prioritize thermal safety and battery longevity, ensuring the device remained safe for continuous skin contact.
THE IMPACT: 99.72% Efficiency & Clinical Readiness
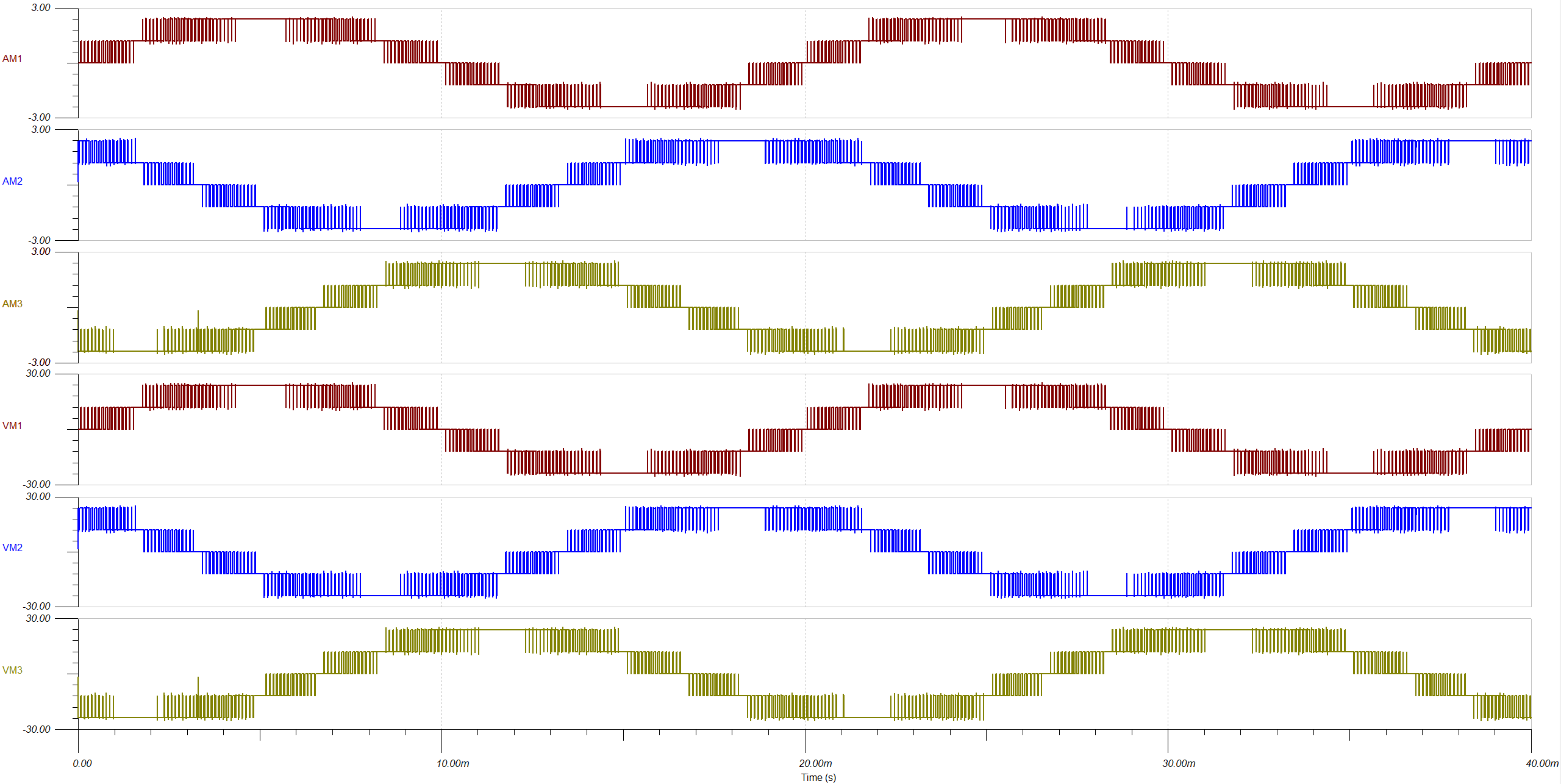
The finalized power system exceeded all clinical and engineering benchmarks, providing a “single-charge” solution for full-day patient mobility:
Peak Efficiency
Achieved 99.72% electronic power efficiency, minimizing heat dissipation and maximizing the 24V, 10Ah battery life.
Weight Reduction
Successfully kept the total prosthesis mass below 2.5kg, directly improving user gait symmetry.
Regulatory Foundation
Documentation from this phase served as the baseline for ISO 13485 design control and Risk Management (ISO 14971) protocols for subsequent clinical trials.




